A Molecule That Can Be Described by Multiple Resonance Forms:
For example molecule benzene has two resonance forms Figure 5. A molecule can have resonance structures when it has a lone pair or a double bond on the atom next to a double bond.

Resonance Structure Easy Science Study Skills Science Chemistry Structure Definition
We can use either of these forms to determine that each of the carbon atoms is bonded to three other atoms with no lone pairs so the correct hybridization is sp 2.

. George Wheland one of the pioneers of resonance theory. A molecule that can be described by multiple resonance forms. We can use either of these forms to determine that each of the carbon atoms is bonded to three other atoms with no lone pairs so the correct hybridization is sp 2.
Rather the actual electronic structure is always the average of that shown by all resonance forms. What are resonance structures. Select the correct answer below.
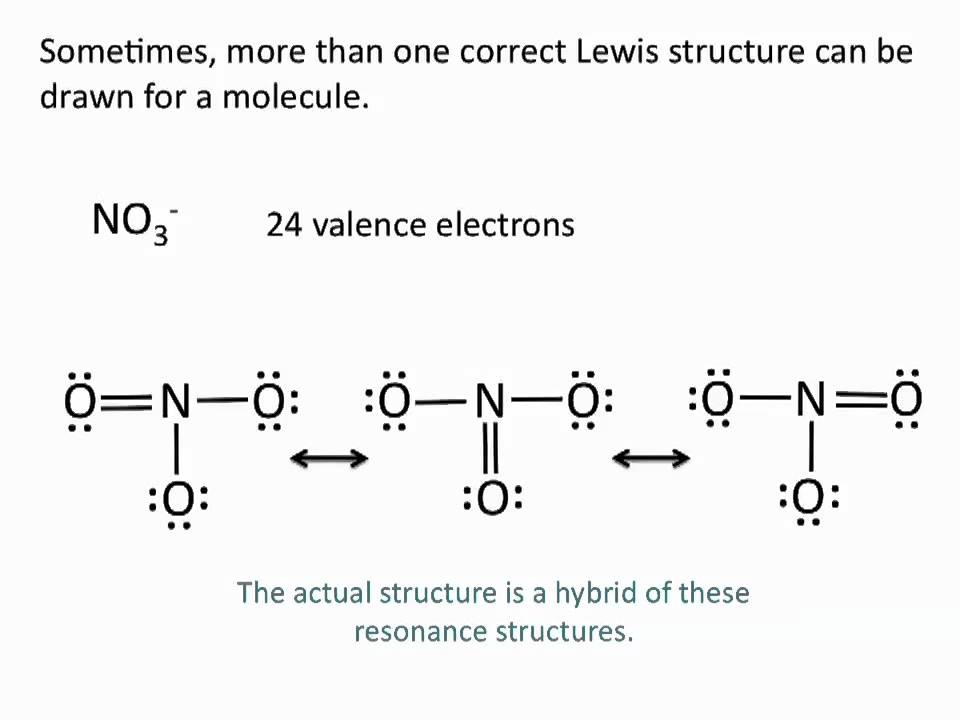
A molecule that can be described by multiple resonance forms. In a resonance structure for NO3 nitrogen has single bonds to two of the oxygens and a double bond to one of the oxygens. Select the correct answer below.
Rather the actual electronic structure is always the average of that shown by all resonance forms. A molecule whose Lewis structure could be described by more than one resonance structure possesses an electronic structure equal to that of a combination of the resonance structures with the more stable structures contributing more to. None of the resonance forms actually exist the reality is the average of them.
Remember that different resonance forms are required to represent the molecule when various arrangements of π bonds are possible. We can use either of these forms to determine that each of the carbon atoms is bonded to three other atoms with no lone pairs so the correct hybridization is sp 2. George Wheland one of the pioneers of resonance theory.
A molecule that can be described by multiple resonance forms. O possesses an electronic structure described by only one of the resonance forms. It does not fluctuate between resonance forms.
In other words the present bond in this molecule can be described from two different configurations. In the first configuration between C and O exists a double bond and the two atoms have zero formal charge. In a resonance structure for NO3 nitrogen has single bonds to two of the oxygens and a.
It does not fluctuate between resonance forms. Neither resonance structure completely describes the electrons. It does not fluctuate between resonance forms.
For example the molecule benzene has two resonance forms. If there are two or more equivalently valid Lewis structures for a given compound these will be called. The electrons in the unhybridized p orbitals form π bonds.
One covalent bond and one partial bond. Whenever you can draw two or more Lewis structures for a molecule differing only in the locations of the electrons the actual structure is none of the structures but is a resonance hybrid of them all. Pg909 The biradical resonance structure for ozone requires two singly occupied MOs and it is clear that an RHF type wave function which requires all orbitals to.
We can use either of these forms to determine that each of the carbon atoms is bonded to three other atoms with no lone pairs so the correct hybridization is sp 2. Arrangement of atoms in a molecule or ion. We should remember that a molecule described as a resonance hybrid never possesses an electronic structure described by either resonance form.
Possesses an electronic structure described by only one of the resonance forms O will rapidly fluctuate in electronic structure between the resonance forms will have an electronic structure that is an average of the resonance forms none of the above. The electrons in the unhybridized p orbitals form π bonds. For example molecule benzene has two resonance forms.
Arrangement of atoms in a molecule or ion resonance situation in which one Lewis structure is insufficient to describe the bonding in a molecule and the average of multiple structures is observed resonance forms two or more Lewis structures that have the same arrangement of atoms but different arrangements of electrons resonance hybrid. Situation in which one Lewis structure is insufficient to describe the bonding in a molecule and the average of multiple structures is observed. For example molecule benzene has two resonance forms.
Possesses an electronic structure described by only one of the resonance forms O will rapidly fluctuate in electronic structure between the resonance forms will have an electronic structure that is an average of the resonance forms none of the above. O will rapidly fluctuate in electronic structure between the resonance forms. For example molecule benzene has two resonance forms.
The other form of elemental oxygen is ozone O3 a molecule that can be represented by the resonance structures. The electrons in the unhybridized p orbitals form π bonds. Two or more Lewis structures that have the same arrangement of atoms but different arrangements of electrons.
The carbon monoxide has two resonance structures limit. Neither resonance structure completely describes the electrons in. Rather the actual electronic structure is always the average of that shown by all resonance forms.
We should remember that a molecule described as a resonance hybrid never possesses an electronic structure described by either resonance form. The electrons in the unhybridized p orbitals form latexpilatex bonds. We can use either of these forms to determine that each of the carbon atoms is bonded to three other atoms with no lone pairs so the correct hybridization is sp 2.
Neither resonance structure completely describes the electrons in the π bonds. Neither resonance structure completely describes the electrons in the. O will have an electronic structure that is an average of the resonance forms.
We should remember that a molecule described as a resonance hybrid never possesses an electronic structure described by either resonance form. Select the correct answer below. A molecule that can be described by multiple resonance forms.
George Wheland one of the pioneers of resonance theory. O none of the above. In chemistry resonance also called mesomerism is a way of describing bonding in certain molecules or ions by the combination of several contributing structures or forms also variously known as resonance structures or canonical structures into a resonance hybrid or hybrid structure in valence bond theory.

Resonance Forms Of Carbonate Mcat Form Molecules
Drawing Lewis Structures Resonance Structures Chemistry Tutorial Youtube Chemistry Science Education Organic Chemistry

Comments
Post a Comment